Abstract
Introduction: Mixed-lineage-leukemia (MLL) is one of the most frequently translocated genes in hematologic malignancies, which produces aggressive leukemias that are refractory to conventional treatment resulting in a 5-year survival rate of only 35%. MLL translocations lead to fusion of the N-terminal fragment of MLL with one of over sixty protein partners, producing MLL fusion proteins that enhance proliferation, block hematopoietic differentiation, and drive leukemia development. Acute myeloid Leukemia (AML) is a heterogeneous and rapidly progressing blood cancer with a poor prognosis, especially in elderly patients where it is more common. Mutations in the nucleophosmin (NPM1) gene are one of the most common causes of AML. Interestingly, the protein-protein interaction between Menin, MLL and MLL fusion proteins plays a critical role in the pathogenesis of acute myeloid leukemias, both with MLLr as well as in leukemias carrying mutations in NPM1. Inhibition of the MLL-Menin interaction with novel Menin inhibitors has shown efficacy in both AML subsets, in pre- clinical and clinical studies. However, the responses are short-lived and hence it is imperative to identify therapeutic approaches that may synergize with Menin inhibition. Our recent work showed that MLLr t(4;11) Acute Lymphoblastic leukemia () patient cells express high levels of the anti-apoptotic BCL-2 protein and are highly sensitive to treatment with the BCL-2 selective antagonist, venetoclax (Benito, J.M., et al , Cell Reports, 2015). Here, we report experiments to establish the efficacy of the novel Menin-MLL inhibitor DS-1594b which was developed by Daiichi Sankyo co., jp, in combination with the BCL-2 antagonist venetoclax (VEN) in MLLr and NPM1-mutated acute leukemias.
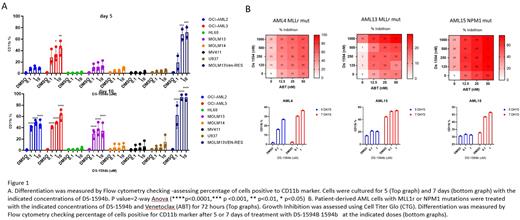
Methods: The efficacy of DS-1594b/VEN combination in MLLr and NPM1mut AML cell lines and primary leukemia samples was evaluated by examining the cell viability in response to serial dilutions of tested compounds as single agents or in combination using Cell Titer Glo (CTG) assay at 3, 5 and 7 days of treatment. Induction of apoptosis was assessed by Annexin V/ staining. Since down-regulation of MLL fusion target genes leads to differentiation in MLL leukemic cells, we monitored cell differentiation by flow cytometry in AML cell lines (MLLr: OCI-AML2, MOLM13, MOLM14, MV411. NPM1 Mut: OCI-AML3 . Leukemia cell lines with no MLLr or NPM1 mutations: HL60, U937) and primary leukemia samples ( blood from relapsed MLLr and NPM1mut AML) using differentiation markers CD11b, CD14, CD15. MLLr cell line MOLM13 resistant to venetoclax was also included in these studies.
Results: 7 days treatment with DS-1594b reduced viability in MLLr and NPM1mut cell lines at low nM concentrations but did not affect growth of negative control U937 and HL60 . DS-1594b induced differentiation in OCI-AML3 and Ven resistant cell after 5 days and in other lines after 10 days (figure 1A). DS-1594b did not induce apoptosis when used as a single agent in any AML cell line tested indicating that differentiation is a key mechanism of Menin inhibition. Five days of treatment with single agent showed a downregulation of Menin in all cell lines and downregulation of BCL2 in OCI-AML3 and OCI-AML2 in Western blot analysis. Remarkably, combination with Venetoclax induced synergistic lethality and growth inhibition in all cell lines. Most importantly, was observed in primary leukemia samples (Figure 1B), with differentiation effects after DS-1594b single agent (bottom).
Conclusions: Here we show in vitro efficacy of DS-1594b, a selective small molecule menin inhibitor, as single agent or in combination with Venetoclax in MLLr and NPM1 mutated cell lines and primary leukemia samples. Preliminary data demonstrate differentiation effects of DS-1594b as single agent in both cell lines and primary AML leukemia samples. Combination effects show synthetic lethality in almost all cell lines except OCI-AML3. The combination elicited profound apoptosis in primary leukemia samples. In vivo studies using MLLr and NPM1 PDXs models are ongoing and will be presented. Overall, the preliminary findings presented here pave the way for further studies aimed at understanding mechanisms of and further development and testing of this combination in AML with MLLr and/or NMP1 mutations.
Disclosures
Skwarska:NOVARTIS: Other: PATENT. Daver:Agios, Celgene, SOBI and STAR Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kartos and Jazz Pharmaceuticals: Other: Data monitoring committee member; Karyopham Therapeutics and Newave Pharmaceutical: Research Funding; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Novartis, Jazz, Amgen, Servier, Karyopharm, Trovagene, Trillium, Syndax, Gilead, Pfizer, Bristol Myers Squibb, Kite, Actinium, Arog, Immunogen, Arcellx, and Shattuck: Consultancy, Other: Advisory Role; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Gilead, Immunogen, Pfizer, Bristol Myers Squibb, Trovagene, Servier, Novimmune, Incyte, Hanmi, Fate, Amgen, Kite, Novartis, Astex, KAHR, Shattuck, Sobi, Glycomimetics, Trillium: Research Funding. Konopleva:Stemline Therapeutics, F. Hoffman La-Roche; Janssen: Membership on an entity's Board of Directors or advisory committees; Reata Pharmaceuticals, Novartis and Eli Lilly: Patents & Royalties; AbbVie, Genentech, F. Hoffman La-Roche, Eli Lilly, Cellectis, Calithera, Ablynx, Stemline Therapeutics, Agios, Ascentage, Astra Zeneca; Rafael Pharmaceutical; Sanofi, Forty-Seven: Research Funding; Forty-Seven; F. Hoffman LaRoche: Honoraria; Stocks, Reata Pharmaceuticals: Current equity holder in publicly-traded company; AbbVie, Genentech, F. Hoffman La-Roche, Stemline Therapeutics, Amgen, Forty-Seven, Kisoji; Janssen: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal